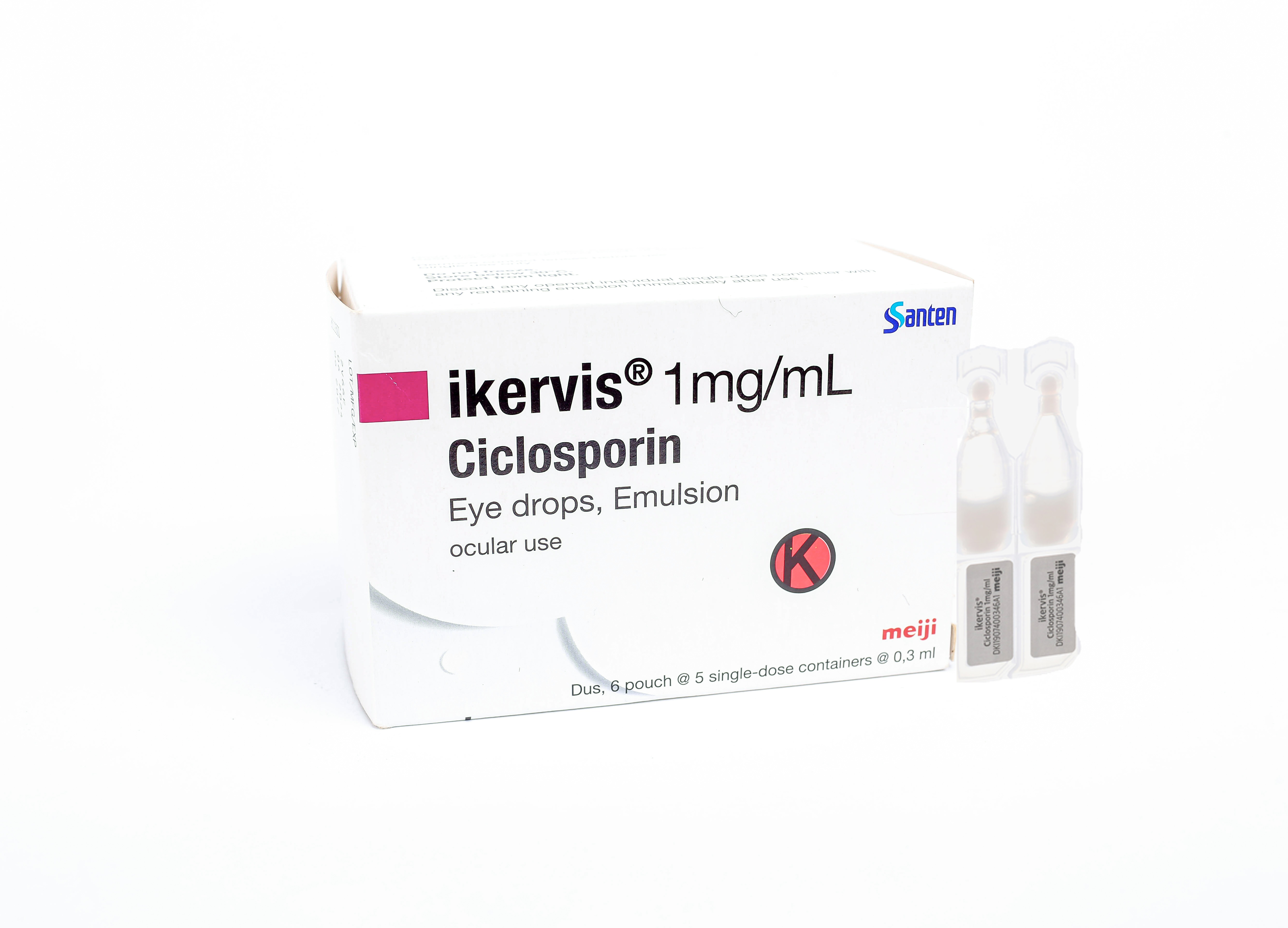
ikervis® Eye drops
ikervis®Eye drops |
|
Composition |
|
Product Category |
|
Indication |
|
Dosage and Administration Posology: 1. Adults The recommended dose is one drop of Ikervis® once daily to be applied to the affected eye(s) at bedtime. Response to treatment should be reassessed at least every 6 months. If a dose is missed, treatment should be continued on the next day as normal. Patients should be advised not to instill more than one drop in the affected eye(s). 2. Elderly patients The elderly population has been studied in clinical studies. No dose adjustment required. 3. Patient with renal or hepatic impairment The effect of Ikervis® has not been studied in patients with hepatic or renal impairment. However, no special considerations are needed in these populations. 4. Pediatric populations There is no relevant use of Ikervis® in children and adolescents aged below 18 in the treatment of severe keratitis in patients with dry eye disease, which has not improved despite treatment with tear substitutes. Method of administration: Ocular use. Precautions to be taken before administering the medicinal product Patient should be instructed to first wash their hands. Prior to administration, the single-dose container should be gently shaken. For single use only. Each single-dose container is sufficient to treat both eyes. Any unused emulsion should be discarded immediately. Patients should be instructed to use nasolacrimal occlusion and to close the eyelids for 2 minutes after instillation, to reduce the systemic absorption. This may result in a decrease in systemic undesirable effects and an increase in local activity. If more than one topical ophthalmic medicinal product is being used, the medicinal products must be administered at least 15 minutes apart. Ikervis® should be administered last. |
|
Contraindications |
|
Adverse Drug Reactions The majority of adverse reactions reported in clinical studies with the use of Ikervis® were ocular and mild to moderate in severity. |
|
Storage After opening of the aluminum pouches, the single-dose containers should be kept in the pouches in order to protect from light and avoid evaporation. Any opened individual single-dose container with any remaining emulsion should be discarded immediately after. |
|
Packaging and Registration Number |