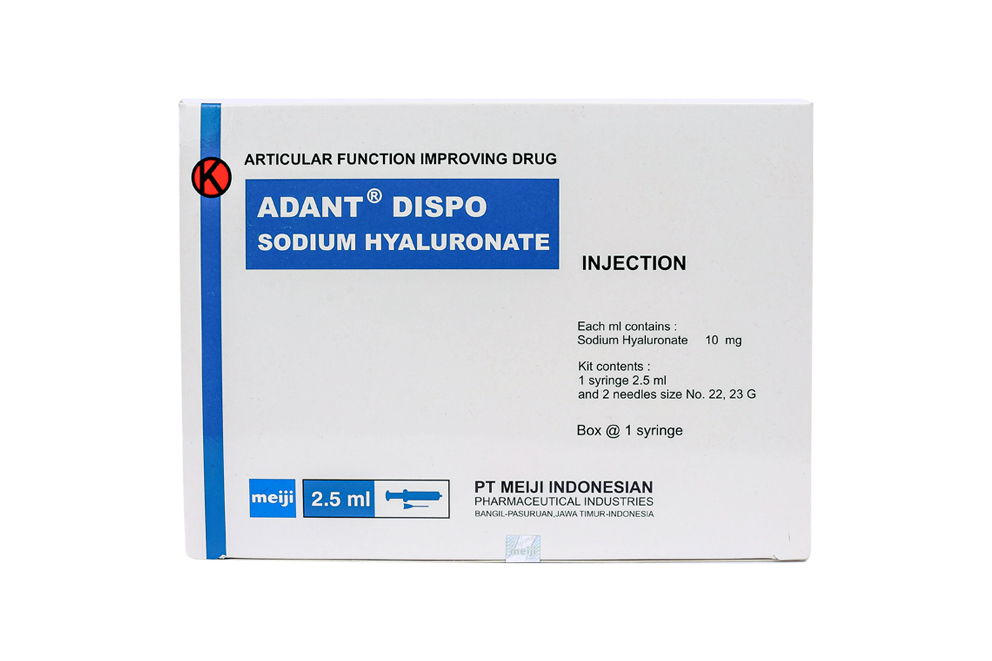
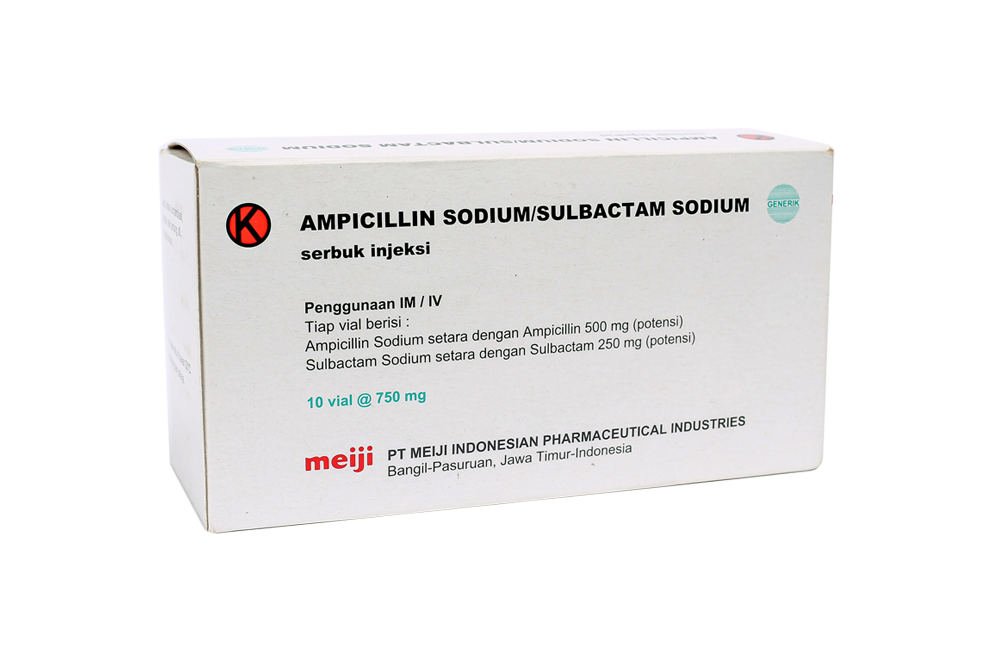
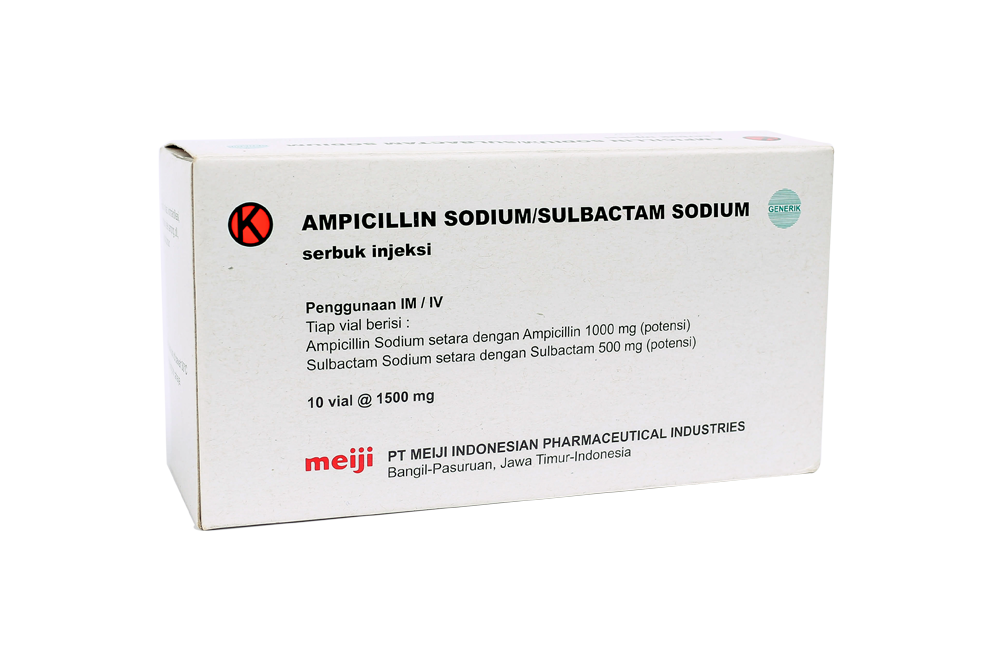
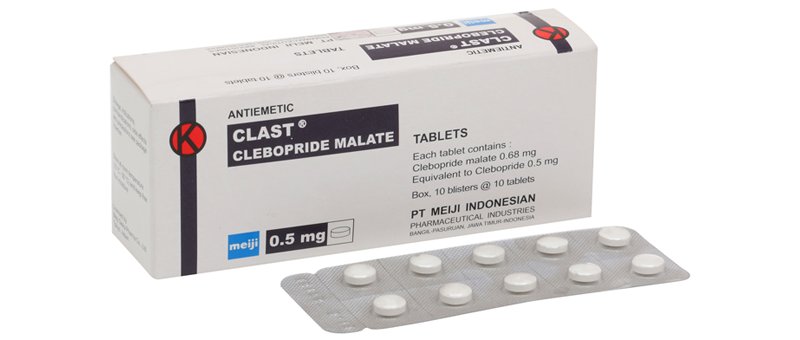
All materials listed on the PT. Meiji Indonesia product list are to provide correct and precise information to website visitors according to their respective backgrounds.
Misuse of activities from the material listed on this Website is the personal responsibility of each visitor. The visitor hereby declares that he is willing to be asked for information by the authorities if necessary and is ready to be processed in accordance with the applicable laws and regulations in Indonesia.
If there are parties who provide information that is not in accordance with the above, PT. Meiji Indonesia is not responsible for the impact caused both to themselves and to the wider community.